A continuous blood pressure monitoring bracelet
My test drive of a promising device
For more than a decade there’s been a relentless effort to develop and validate a continuous (or more precisely continual), non-invasive, cuff-less, accurate, blood pressure device—a holy grail in sensor technology. I’ve been testing one by Aktiia this week (my results from yesterday below), which is not yet available in the United States, and have made multiple side-by-side comparisons to an Omron standard cuff and the results have correlated well. I don’t have hypertension and by no means am I ready to say this device is ready for prime time, but it has many useful and distinctive features.
It’s a very lightweight bracelet that has an optical sensor which, after a traditional arm cuff calibration (done once a month), transforms the photoplethysmography (PPG) signals into blood pressure estimates using pulse wave analysis algorithms. The nice part is that a single charge for the sensor lasts about 2 weeks, so it’s great for wearing at night to get 24/7 data.
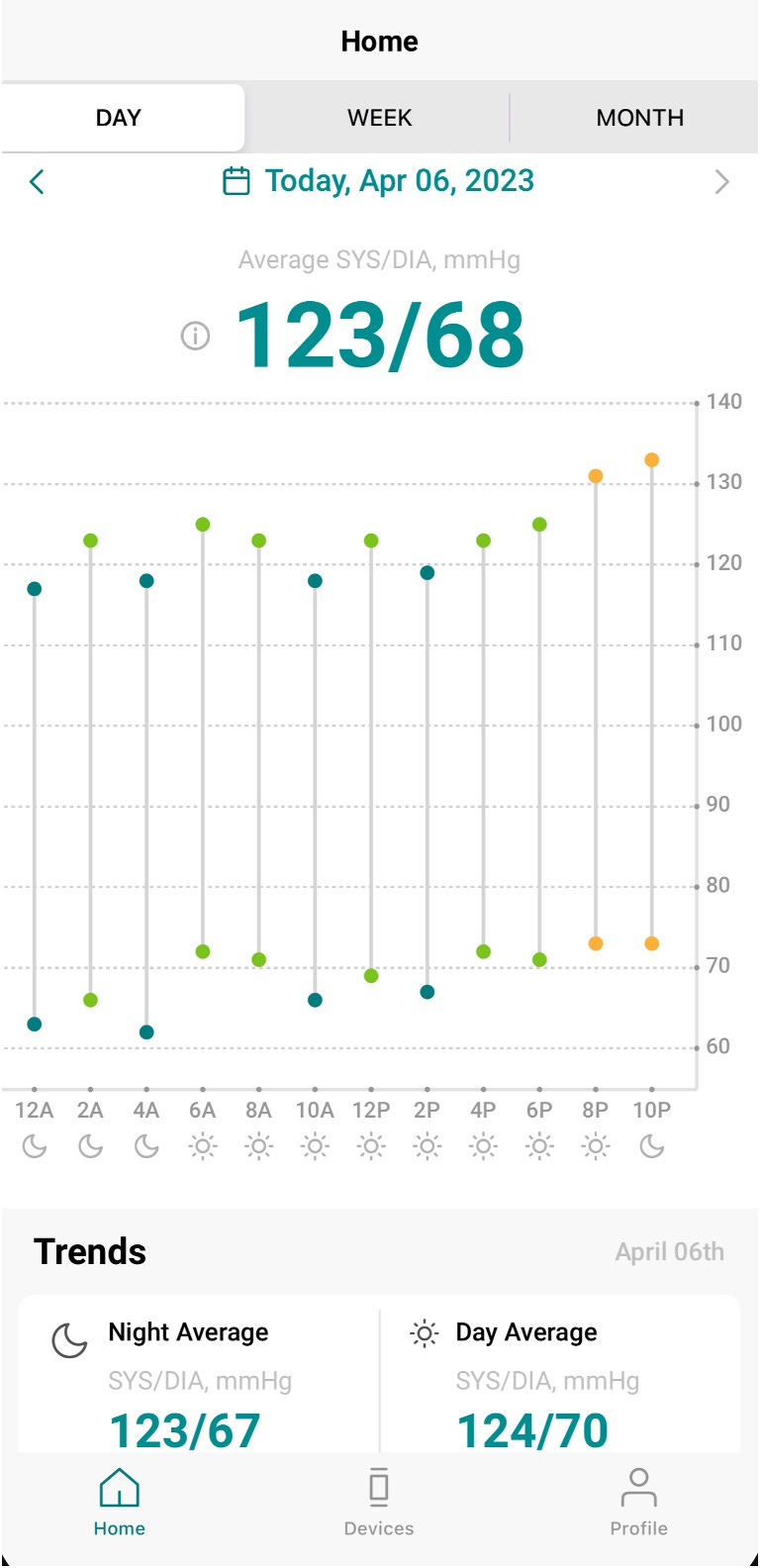
Data capture and display require Bluetooth pairing with a smartphone and Internet connectivity for synchronization with the company’s cloud server. The passive, automated average BP data (from measurements triggered hourly) are presented for each 2-hour period as shown in my recordings from yesterday. Of note, if you’ve ever tried an ambulatory BP arm cuff device, it doesn’t work well for night readings as the inflation wakes you up.
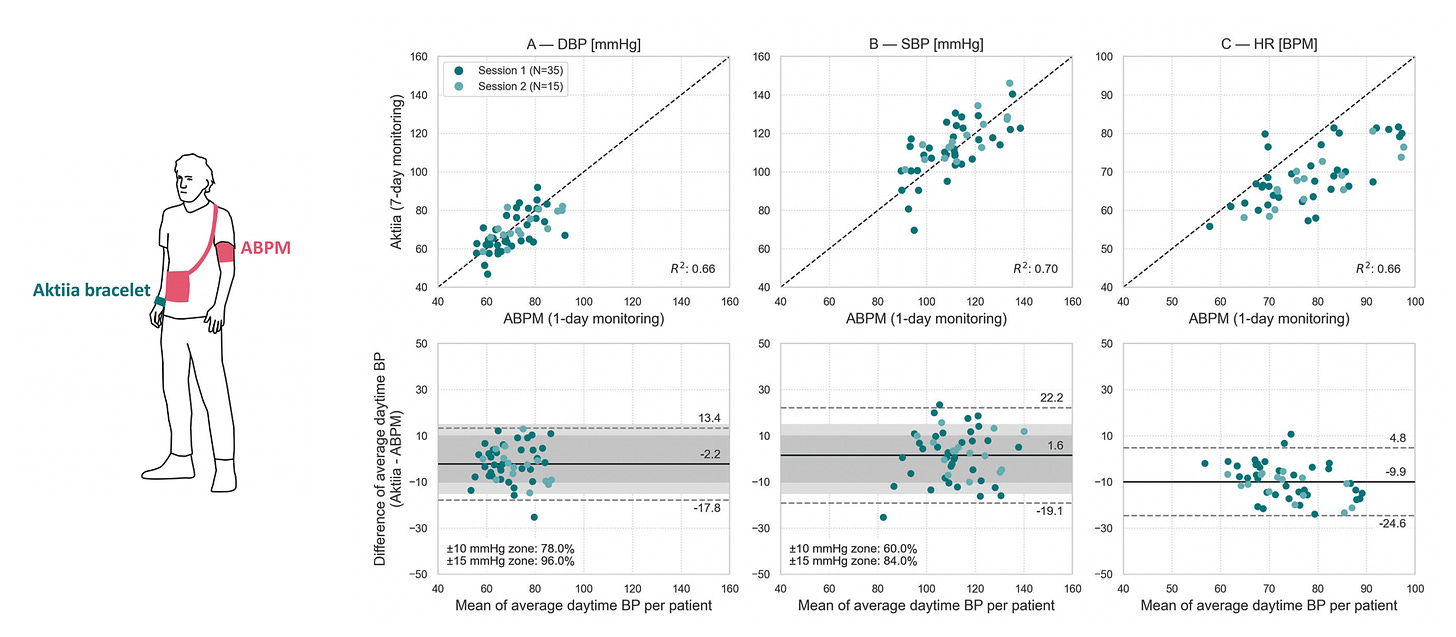
The Swiss company that makes this device has multiple peer-reviewed publications assessing it such as here, here, here, and the most recent one here. That recent study was a comparison of traditional ambulatory arm cuff measurements during the day versus the Aktiia device in 52 participants for 1 week. The device also senses heart rate (HR) and the 3 panels below show reasonable correlation between diastolic blood pressure (DBP), systolic blood pressure (SBP) and heart rate. You will note that the upper end BP measurements (for Aktiia’s algorithmic determinations these would be better described as estimates) were for systolic below 160 mm Hg and diastolic above 50 mm Hg. Aktiia says it measures BP between 0 and 299 mm Hg (and heart rate 40-199 bpm), but I haven’t seen data from their reports that is in the extreme range in either direction. The correlation with a traditional BP cuff device has been shown to be good: The standard deviation of the averaged paired differences per individual was 6.55 mmHg for SBP and 5.15 mmHg for DBP. The bracelet has memory to store 30 days of sensor data. A limitation is that you can’t get a spot measurement when you're anxious or upset, and that BP readings are only obtained when there is no movement (not during exercise). A new independent report out today in 41 participants suggest there is an issue for night time BP decline accuracy, which goes along with the lack of any significant dip seen in my recordings.
All of the publications are reporting a limited number of participants (<100 in each) and have similar issues with the BP range assessed, and except for the one cited about night BP potential inaccuracy they are all company reports, not by independent researchers. The performance with dark skin pigmentation (with increased absorption of light) and high hair follicle density (a potential optical barrier) has been assessed, and is reassuring and fulfilled the ISO (International Organization for Standardization) standards, but only evaluated in a limited number of people. The device has European commercial approval since December 2020, CE-mark (Class IIa), and was compared with invasive arterial line measurements in patients lying in bed in an intensive care unit and vs individuals sitting using traditional auscultation methods. It has not yet been cleared or approved by the United States FDA.
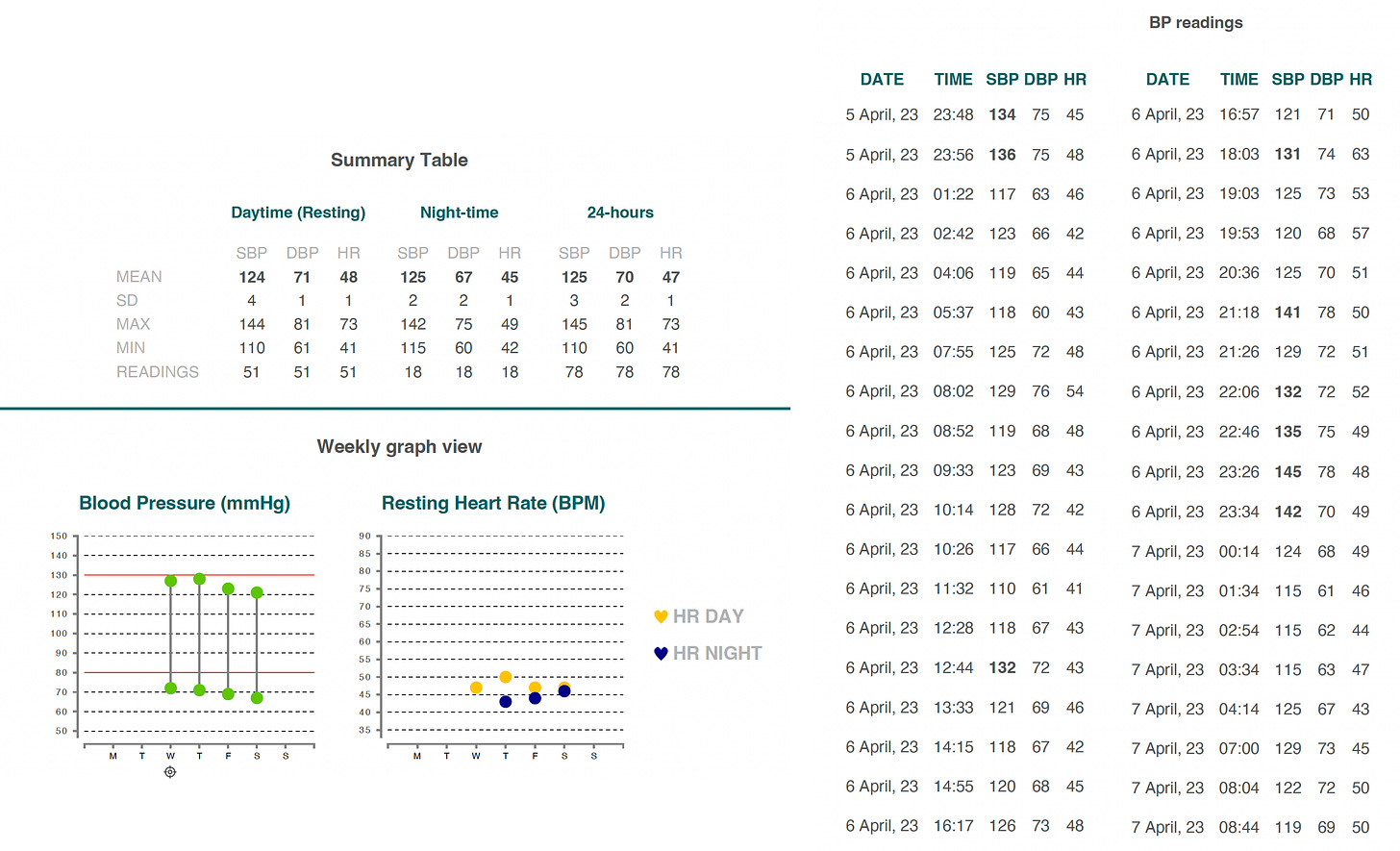
The device data generates a report with all the readings and the summary, as shown below for me, that can easily be shared with a physician.
Where available in the UK and many countries in Europe, the device costs 199 Pounds, 230 Euros in France, Germany, Austria and Italy which would be just over $250 in the US, assuming the price doesn’t differ.
Bottom Line
The Aktiia device has considerable promise for a number of reasons. It is automated, easy to use, provides a lot of data throughout the day and night (which helps diagnose whether there is the expected night time dip) and has had multiple assessments with publications. As a device with continual, automated BP readings, it is altogether different from BP devices reviewed in this NY Times piece from late last year. The main rub is whether the device will prove its accuracy, such as with extremes of BP in large scale assessments. It will likely cost at least 2 to 3-fold that of the devices on the market that include wireless BP cuffs that have nice smartphone data visualization. I remain concerned about relative lack of diversity of the research participants, and that as we have seen with pulse oximetry which relies on an optical sensor, there could be a problematic bias of results in people of color. Nevertheless, I suspect it will get FDA clearance in the months ahead, so it will become an option.
Hypertension is the most common chronic medical condition, with half of American adults affected, and more than half of those who do not have adequate control, leaving them more vulnerable for adverse outcomes. In clinic this week I saw 3 patients who brought in sheets of paper with manual recordings of their blood pressure (during the day) and they are the exceptions—the people who are willing to take the time and obtain the readings and record them. The smartphone summary of data will help people manage their BP better, knowing from the multiplicity of measurements if and when their BP is not adequately controlled (mostly too high, but sometimes too low, i.e. over-medicated). As a physician, the data reports shared from patients using such a device would be extremely helpful towards achieving optimal patient BP management. It is very hard to get patients with hypertension to take multiple readings per day, and impossible during the night. The night BP elevation could signify sleep apnea (or other sympathetic nervous system dysfunction) which might otherwise be missed. If the lack of accuracy of night time decline in BP readings turns out to be replicated, this would be an important concern. Notwithstanding this issue, the frequency and 24/7 breadth of data is something we have wanted for such a long time so it’s exciting that we may be approaching the time when it’s possible. I look forward to more extensive evaluation of this device and others that may ultimately fulfill our dream of cuff-less, automated, accurate BP measurement. And that such sensors could be integrated with smartwatches to avoid the need for more than one wrist device.
Thanks for reading, sharing and subscribing to Ground Truths. As I’ve previously announced, if you decide to become a paid subscriber all proceeds will be donated to Scripps Research.
N.B. I have no relationship with Aktiia, the device reviewed here. I have no grants or any financial connects with the company. The device was recommended to me to try by a colleague and I was able to get the TestFlight app from the company via email to go ahead with my testing.