“In the medicine of the future, molecular physicians built from a patient’s own cells might ferret out cancer, repair injured tissue, and even remove plaque from blood vessels.”—Elizabeth Pennisi, Science Magazine
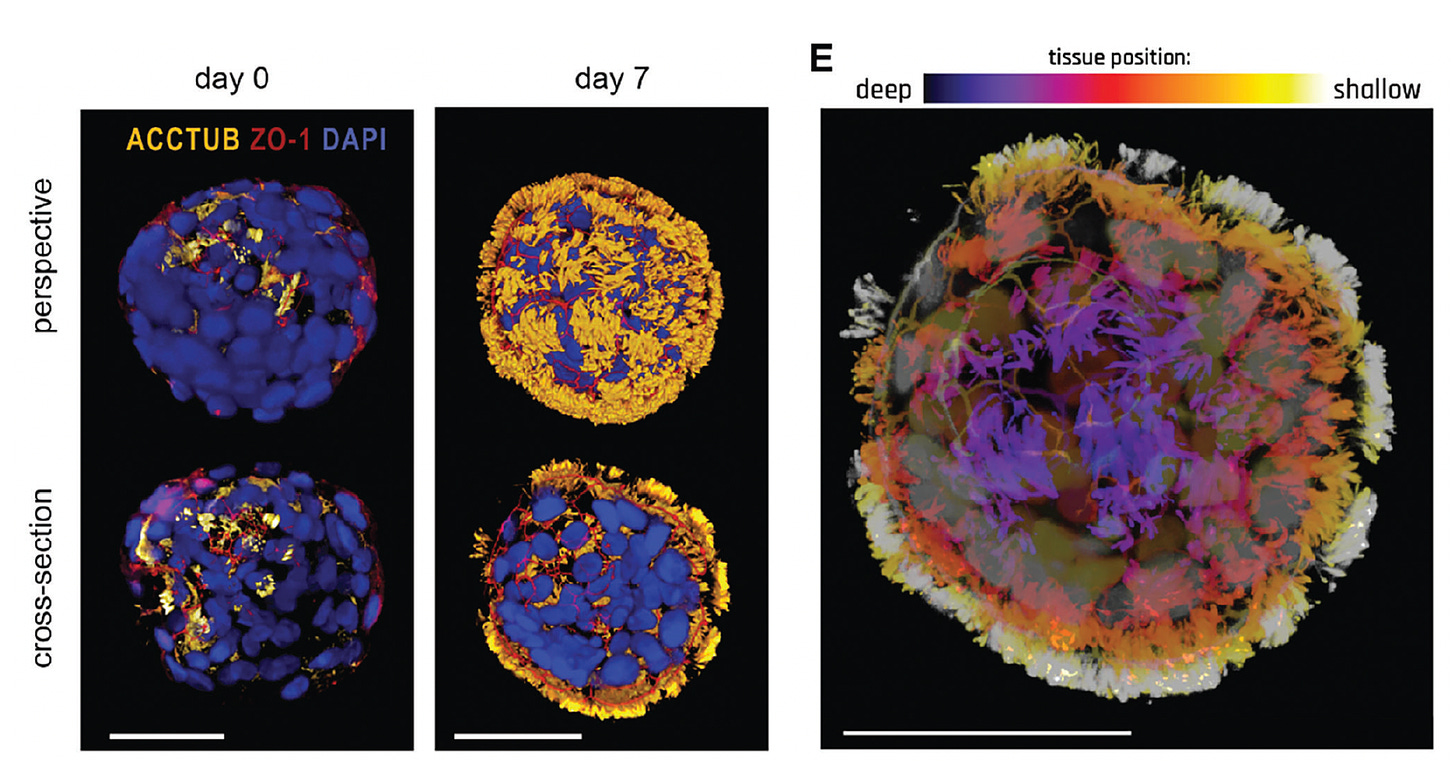
That’s quite the lede about a striking paper that came out this week. It seems like science fiction, but it’s not. It’s about the Tufts University group, led by Michael Levin, discovery of human biobots—cells that self-organize to become multicellular and mobile—as seen below, derived from human lung (trachea) cells that had been laying flat in culture for decades. Previously mobile biobots from frogs (xenobots) had been demonstrated by these investigators, but this is the first report to show this capability from human cells, no less the potential to repair tissue.
Their motility (the big moving cell is the biobot or, as they called it, Anthrobot) is derived from the cilia inside trachea cells that were turned inside out by immersion in a special, less viscous bath, as seen below in this video.
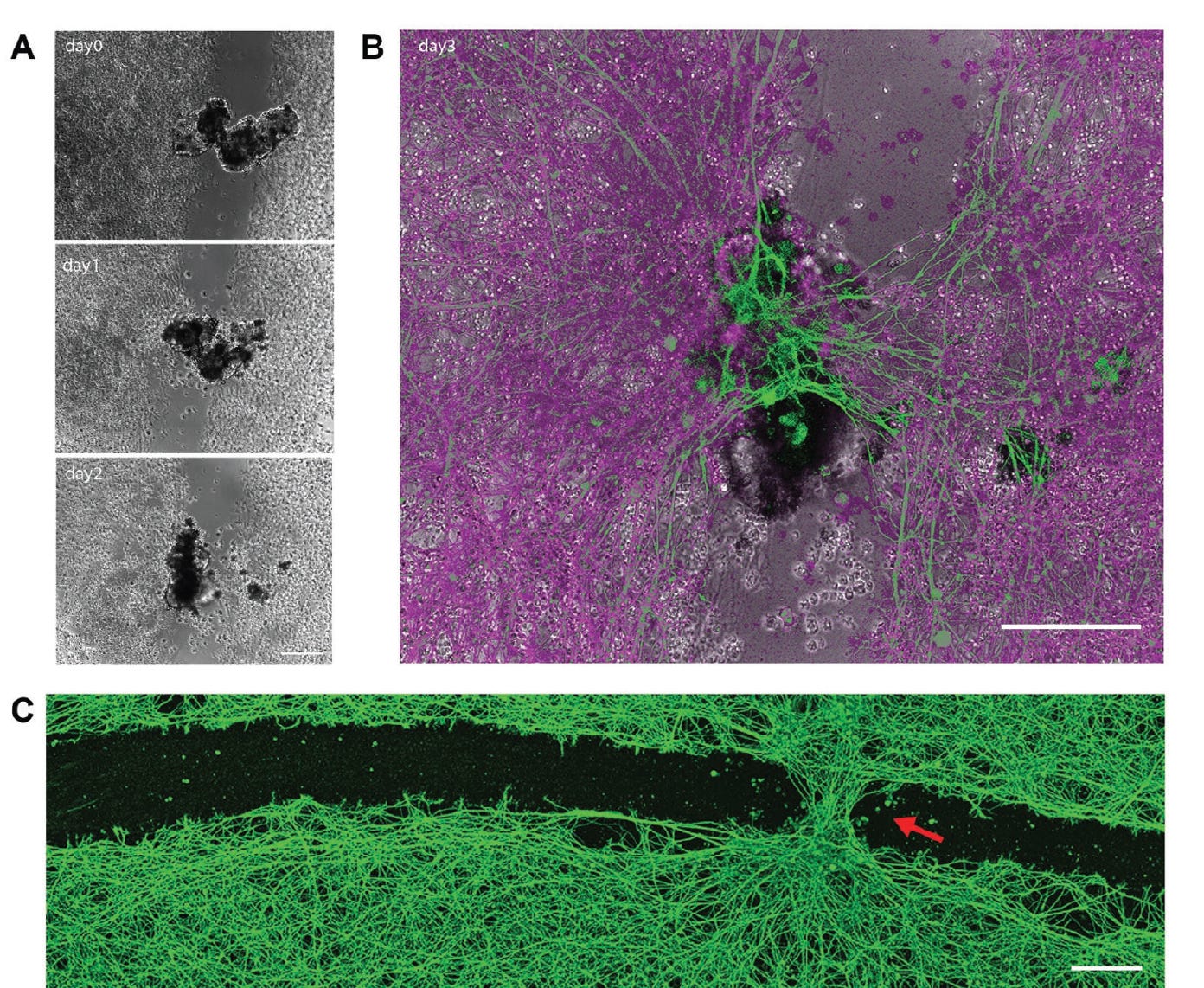
When these cells were placed on a layer of neurons with a scratch made to simulate injury, surprisingly, the biobots attempted to repair the gap (video below). The biobot is traversing the scratch gap, initiating the fix-up. Surprisingly— since this was autonomous, as the authors point out “a repair property that likewise could not have been guessed in advance from existing frameworks describing the uses of organoids and other bioengineered structures.”
By day 3, the biobots had created a stitch repair of nerve tissue as seen below
Background on Organoids
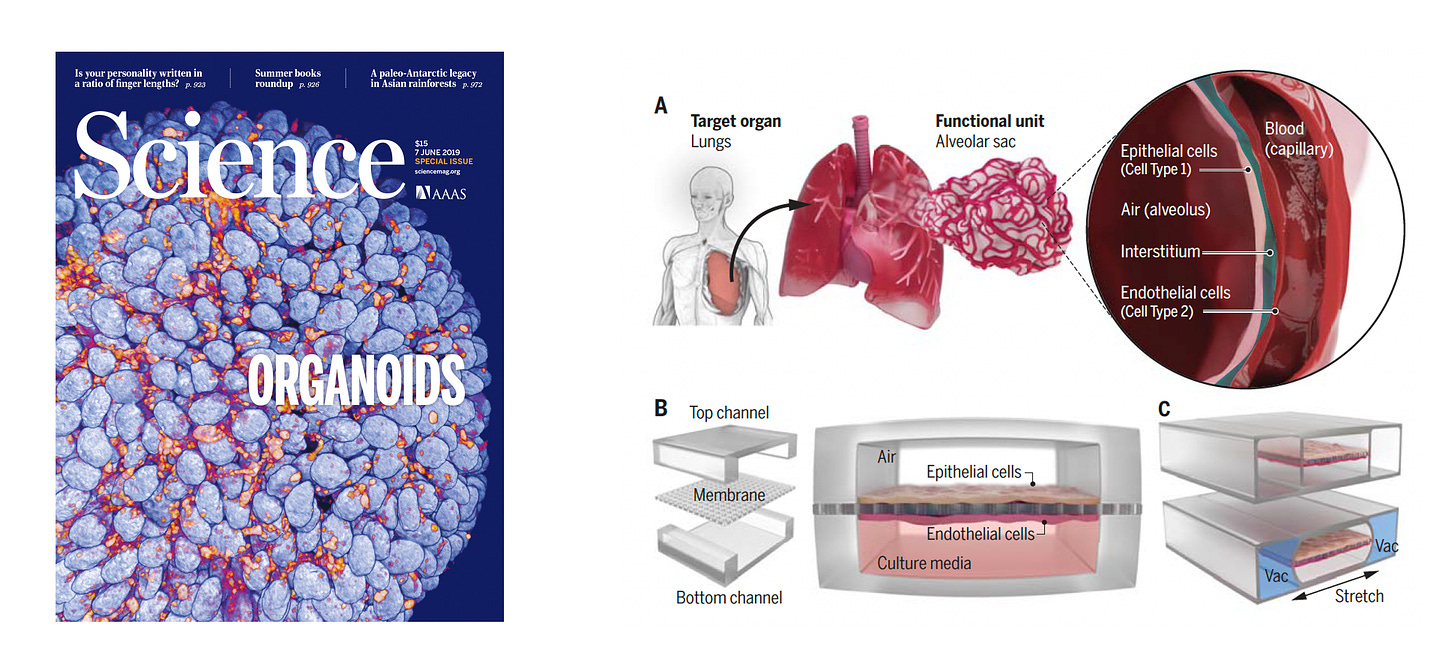
Organoids are attempts to mimic an organ using cells that are cultured in 3D. They are usually derived from induced pluripotent stem cells (iPSCs, from our white blood cells or skin), embryonic cells, or adult cells; the new biobot report used normal adult bronchial epithelial cells. Organoid models of most organs have been produced including brain, retina, pancreas, heart, liver, intestine, and kidney. The cells have an extraordinary ability to self-organize when placed in a 3D extracellular matrix to mimic many of the properties of the organs. For example, brain organoids have been shown to have electrophysiologic (brain wave) properties. Models to simulate various diseases, including Alzheimer’s and Parkinson’s, have been developed, along with testing drug interventions in culture, and CRISPR genome editing to alter cell gene expression and functionality. These “organ-in-a-dish” constructs have helped understand how organs develop and the diseases they intend to model. While they have progressed substantially in recent years for approximating structure and functionality of human organs, there still is a large gap. We’ve even seen “organoids-on-a-chip developed to reduce the gap by controlling the microenvironment, addressing tissue-tissue interactions and reducing variability (below, right panel)
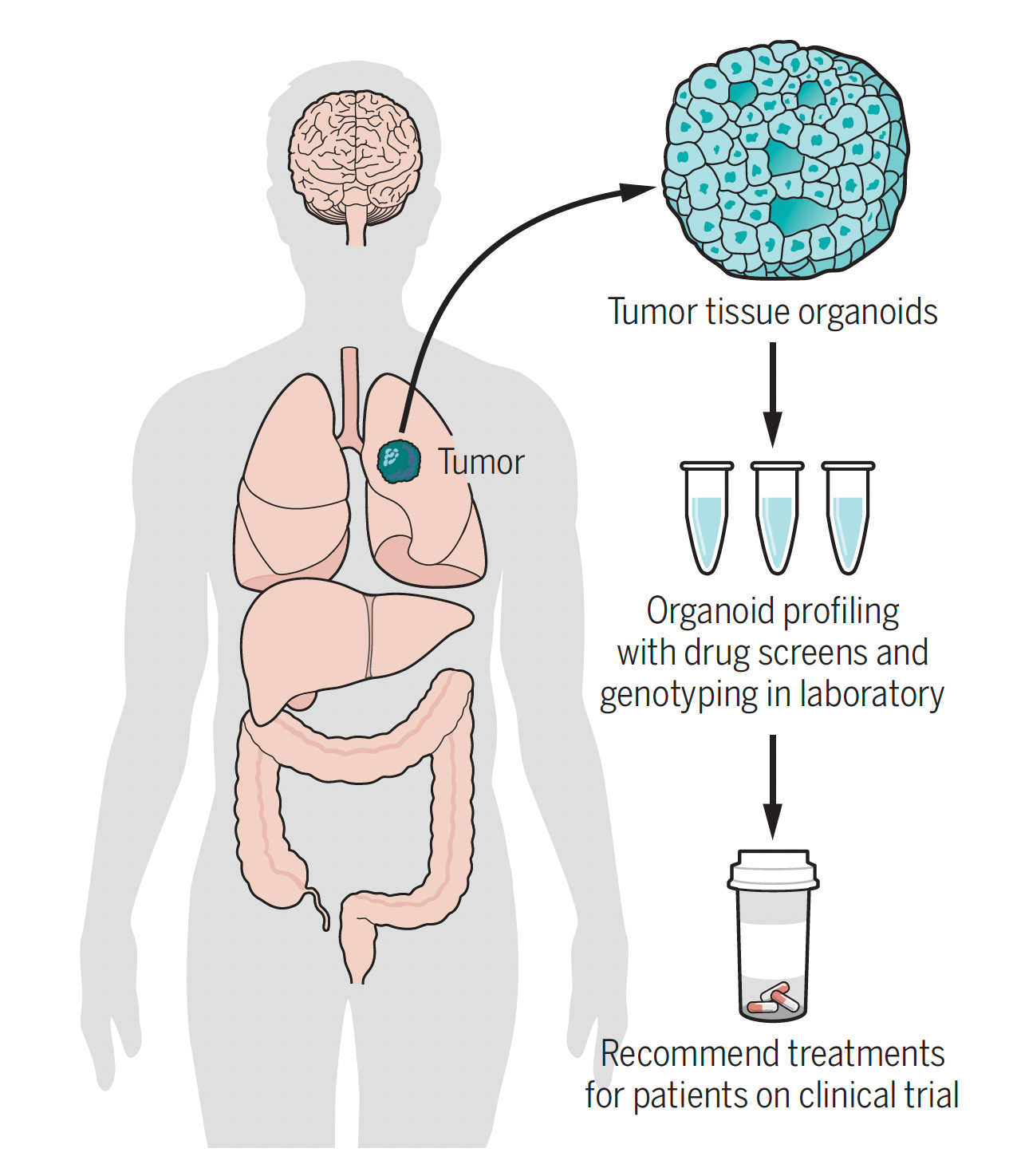
Considerable work with organoids has been done to simulate cancer, and this has begun to reach the clinic for predicting an individual’s response to therapy using patient-derived organoids (PDOs), as schematically shown below.
The First Cardioid
This week was also marked by the first report of a multi-chamber heart organoid called a cardioid. This is being used to elucidate congenital heart diseases—the causes of which still and remarkably remain unknown. The importance of the first multi-chamber heart organoid is that there is much chamber-specific gene expression, embryology, and differential effects of teratogens (drugs or toxins inducing mutations).
Significance
The new human biobot report is unique, since it represents the first time the role of organoids has morphed from studying a disease or determining the success of a drug intervention to actually using the organoid product as a repair tool. As imagined in the conclusion of the paper:
“In the future [Anthrobots] could be personalized for each patient, enabling safe in-vivo deployment of these robots in the human body without triggering an immune response. Once inoculated in the body via minimally invasive methods such as injection, various applications can be imagined, including but not limited to clearing plaque buildup in the arteries of atherosclerosis patients, bulldozing the excess mucus from the airways of cystic fibrosis patients, and locally delivering drugs of interest in target tissues.”
We’re obviously a long way from those diverse and far-reaching potential use cases. The report needs to be independently replicated. It’s only in vitro—in the lab, in a dish. We’ve already seen how creation of organoids is laborious, takes time, depends a lot on composition of culture media, requires substantial expense and resource consumption, all pointing to a lack of scalability. That’s one of the reasons organoids for predicting a patient’s response to therapy for cancer hasn’t yet been used much.
Nevertheless, the discovery of cells that can autonomously repair tissue distinct from their origin is exciting and opens up potential for a new era of tissue engineering that previously hadn’t even been conceived. Moreover, as we’ve seen with T-cell engineering, we can genome edit cells before they are administered back to a person. That would make for edited biobots, likely another refinement in the works. While recently there’s been fascination with how quickly generative A.I. GPT chatbots are progressing, there’s now a parallel path potential with biobots.
Thanks for reading, sharing and subscribing to Ground Truths!
All content from the Ground Truths newsletter posts and podcasts are free.
Any and all voluntary financial support goes to Scripps Research.




Thank you for bringing to our attention, yet once again, an extraordinary medical advance that is on the horizon. This potential application really caught my eye: “various applications can be imagined, including but not limited to clearing plaque buildup in the arteries of atherosclerosis patients.” If I am understanding this correctly, could this be a way to reduce the number of people on lifelong medication regimens to address this problem? If so, that appears to me to offer a huge a benefit.