The bivalent vaccine booster outperforms
A review of the cumulative body of evidence
You may recall that I was a skeptic about the bivalent BA.5 vaccine when the FDA gave it the OK to roll out in September without any human data. At the very least, I had hoped there would be lab studies to confirm a strong immune response to this variant, and that it was superior to the original (monovalent) booster that was directed to the ancestral (Wuhan) strain. But we now have extensive data that is quite encouraging—better and broader than expected— that I’m going to briefly review here.
Lab Studies
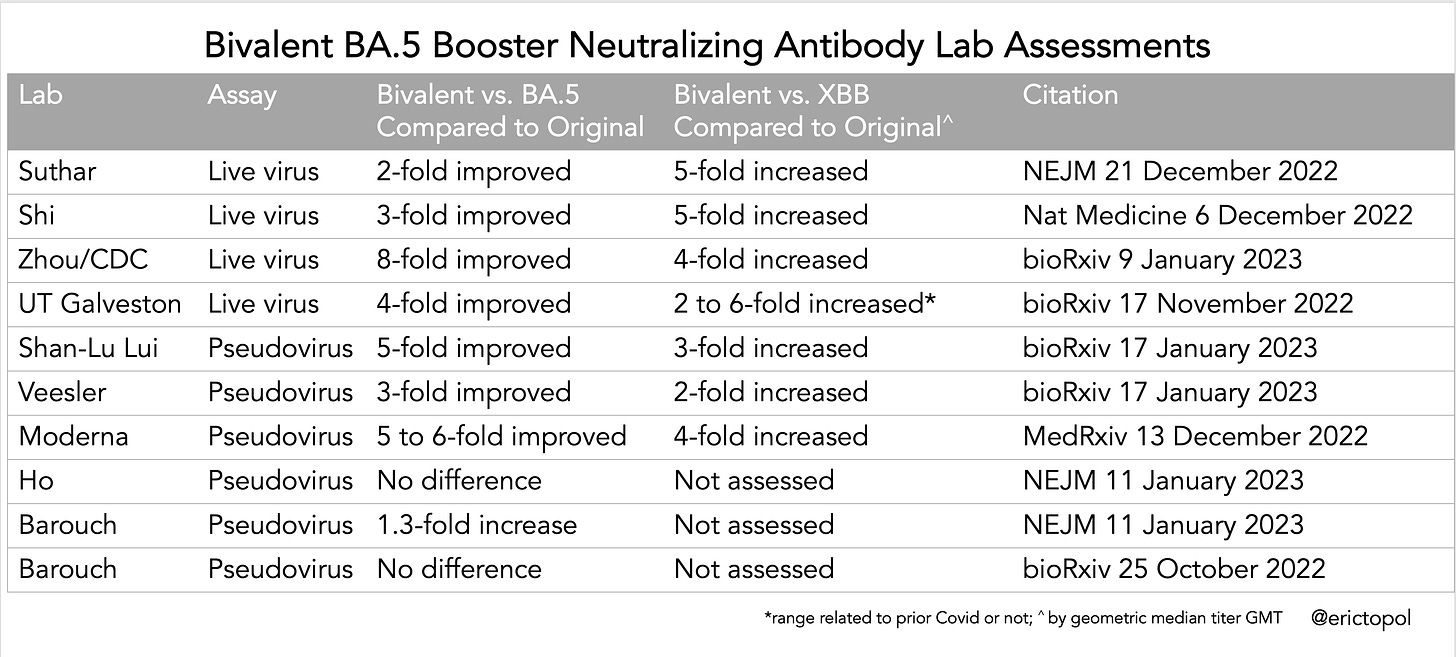
These are assessments for the neutralizing antibody response to the bivalent (BA.5 directed) booster compared with the original (Wuhan strain directed) booster as a function of variant. There have been 10 such studies. Of the 8 reports, 4 used live virus and 6 used pseudovirus assays. Overall, these showed consistent evidence (7 of 10 reports) of the bivalent’s superior neutralizing antibody response to BA.5 (compared with the original booster) as hoped and anticipated, but also against XBB, which is fortuitous since XBB.1.5 is on a rapid path to US dominance now, having already reached >70% in the Northeast region. This is indicative of broadened immunity against other new variants that were induced by the bivalent.
The new Israeli study
Among more than 700,000 participants age 65 and older, who either had the bivalent or did not, there was an 81% reduction of Covid hospitalizations (Figure below) and an 86% reduction of deaths. During over 70-days follow-up, there were 73 deaths in those without the bivalent and 1 death of a participant who received it (among ~622,000 and 85,000 seniors, respectively).
The CDC report
On December 30, 2022 the CDC issued 2 Morbidity and Mortality weekly reports (MMWR) on the initial bivalent booster vaccine effectiveness. Here are the data for age 65 and older, showing marked protection vs. hospitalization ranging from 73 to 84%, comparing not only to those unvaccinated but also those individuals who had been vaccinated and boosted. A second report was for all adults age 18 and up, showing bivalent booster 38 to 57% effectiveness vs hospitalization, which is consistent, but of lower magnitude than for seniors. This would be expected in a lower risk group with a high incidence of infections in the unvaccinated controls. It is notable how well the CDC data for seniors lines up with the Israeli data above.
CDC Results Through November 2022, the BQ.1.1 wave
On December 28, 2022, the CDC posted updates data for the bivalent vaccine protection vs hospitalization. During November, for people age 65 and older, there was a 90% reduction of hospitalizations for the bivalent vs unvaccinated, a 13.5-fold increased risk of hospitalization for the latter group, and 2.5 fold higher rate among seniors vaccinated but without a bivalent booster. The relative reduction was similar for all adults age 18+, albeit with less absolute benefit in the overall adult population of younger age groups at lower risk. The consistent and large reduction of hospitalizations, especially among the vulnerable during the BQ.1.1 wave, reflects the broadening of the immune response for the bivalent as compared with the original booster shot
The Bivalent Vaccine Detractors and Naysayers
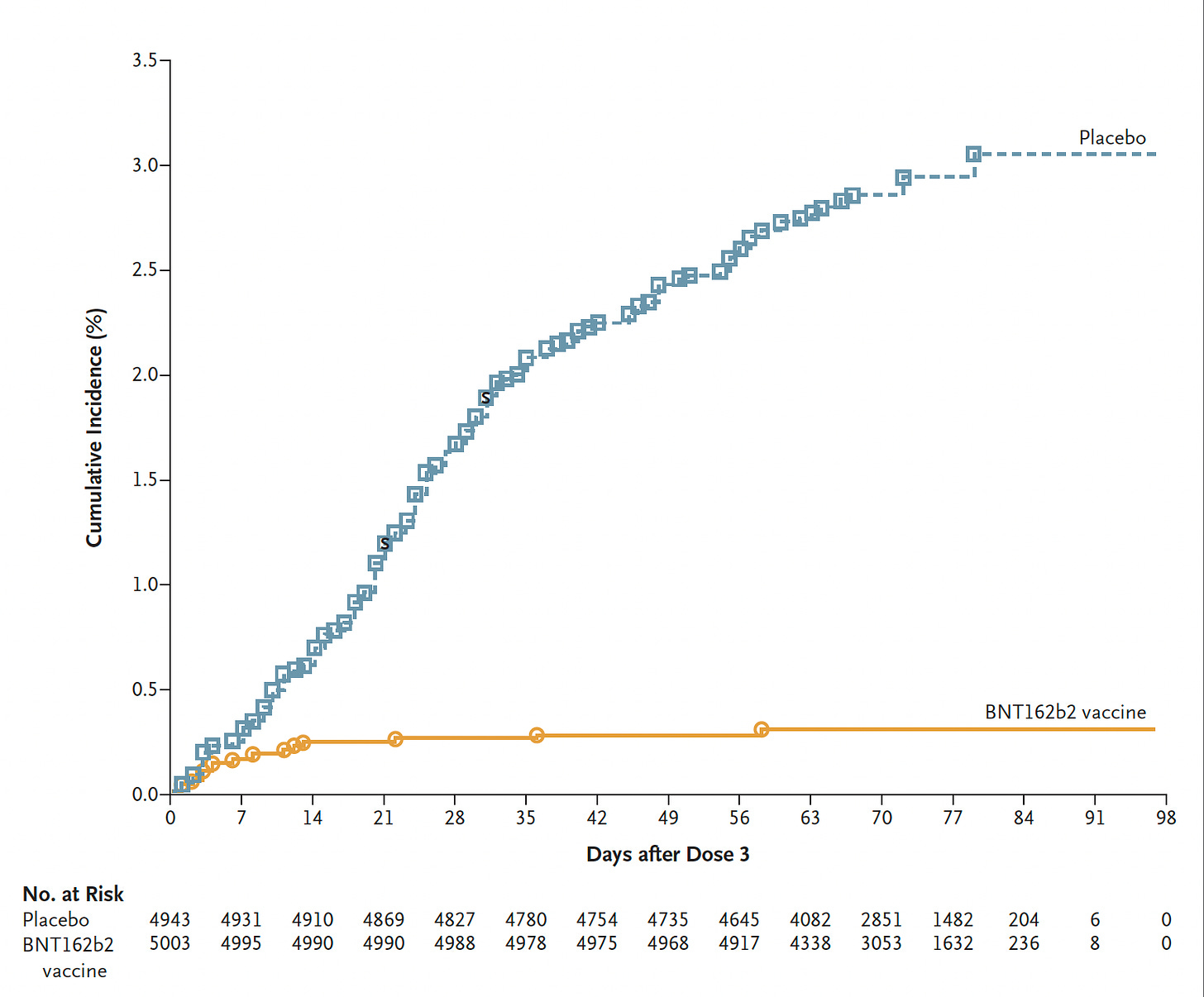
The FDA and the United States government made a big bet on the bivalent BA.5 boosters with a purchase of over 170 million doses, without direct human data, extrapolated from some limited clinical data with the BA.1 bivalent, mouse experiments, and the influenza vaccine experience. The acceptance and uptake of the booster has been poor (only 16% of those eligible, only 38% of seniors), partly due to pandemic fatigue, the reactogenicity of the shots, the delusion that the pandemic is over, and another factor—an outpouring of detractors. This has ranged from editorialists at the Wall Street Journal claiming the boosters induce variants (They don’t. Infections do), to Paul Offit writing opeds (“CDC Oversells the ‘Bivalent’ Covid Shot”)and today’s NEJM piece entitled “Bivalent Covid Vaccines—A Cautionary Tale.” Dr. Offit has been staunchly against boosters since their need—to ante up to waning immunity and risk of severe Covid— was firmly established in the summer of 2021. He constantly negates much of the data, including a randomized trial of nearly 10,000 participants of all ages 16+, that showed the original booster (3 shots vs 2 shots) led to marked reduction of Covid infections (Figure below), and multiple effectiveness studies from countries throughout the world—Israel, UK, Qatar, the US—that unequivocally show the reduction of hospitalizations and deaths with a booster shot during the Omicron waves. Early on I agreed with Offit on some of the cautionary points that he made, but clearly there’s far more data and evidence now that he has chosen to disregard.
There is now more than ample, highly consistent evidence via lab studies and clinical outcomes to support the bivalent’s benefit over the original booster. There have been demands and calls for randomized trial comparing the original versus the bivalent booster. Randomized trial takes time to set up and would come at considerable added expense. Furthermore, they’re not necessary since there is nothing to suggest any inferiority of the bivalent—all of the data points to added benefit. When the BA.5 bivalent was given FDA approval to roll out, this was done to catch up with the marked evolution of the virus for which there had not been an updated vaccine since the original version designed in January 2020. I’ve been a big supporter and leader of randomized trials throughout my career, but fully appreciate that there’s a time and place when they are appropriate—it’s not in the midst of a pandemic with all the priors (randomized trials and effectiveness studies) that we have now. I disagreed with the FDA decision at the time, but have comprehensively reviewed the data since and believe they (Dr. Peter Marks and colleagues) made the right call.
While the clinical benefit of reduced hospitalizations and deaths is best seen in the high risk population of age 65 and older, there is certainly evidence for benefit across all adults, as reviewed above. That’s the story of subgroup analysis, such that one can pick a subgroup and claim it only works in this demographic, even though there is no indicator of an interaction by subgroup effect. That is to say the totality of data supports broad benefit, irrespective of age.
A key question is how long the bivalent will help prevent severe Covid. There’s no reason to think it would differ much from what we’ve seen with the original booster, with diminished protection over the course of 4 to 6 months. We can’t keep relying on such frequent shots (2-3 times per year) in the future, which is why it is so critical for an enrichment of our preventive armamentarium—more durable vaccines by tweaking nanoparticles, striving for variant-proof efficacy, and mucosal immunity, no less more drugs beyond Paxlovid that can rapidly inactivate the virus. As I recently wrote, The virus isn’t done evolving. It’s talking to us and we’re not listening
The Bottom Line
Bivalent boosters work well to prevent severe Covid, as manifest by reduction of hospitalizations and deaths. They are not a panacea, by any means—their efficacy against infections is limited and of short duration, which has been the case for shots since the Omicron variant came along in late 2021.
The spike protein of the BA.5 bivalent more closely resembles XBB.1.5 than the Wuhan ancestral spike. The lab studies with live virus assays are strongly supportive. The clinical data are unequivocal. While it would be far better to have a “universal” pan-β-coronavirus vaccine that worked against all SARS-CoV-2 variants, and nasal vaccines to help block infections, the bivalent BA.5 booster has helped to bridge the big antigenic distance gap from the Wuhan strain to current Omicron subvariants. We’re lucky in the United States to have a big supply and it’s frankly disappointing to see the divisiveness, cherry picking of data, and detractors that defy the body of evidence that has now accumulated. The bivalent booster impact of broadening our immune response has exceeded expectations.
Addendum re: Possible signal of risk for bivalent in age 65+
Today (Friday the 13th!) the CDC raised awareness of a possible stroke risk in some data they are tracking from a number of US jurisdictions for the Pfizer bivalent booster in people age 65 and over. Importantly, this is an isolated finding not seen in any other dataset, including the Medicare database, the Veterans Affairs, the FDA, VAERS, Israel, the UK, or Europe. Moreover, it has not been seen with the Moderna bivalent, which uses a significantly higher dose of mRNA. It is certainly good to see such close monitoring by CDC. The possible signal is certainly worthy of continued assessment but based on the data I have seen it does not warrant concern and is likely a spurious finding based on a limited sample without corroboration by any other—and much larger—dataset.
Thanks for reading, subscribing and sharing Ground Truths.
Wishing you a healthy and fulfilling 2023