The Virus Takes a Detour in its Evolutionary Arc
How Well will Our Immune System Respond?
Two recent reports of population immunity pressure (here and here), from prior infections and vaccinations, have concluded this is the principal driver of recent SARS-CoV-2 evolution. As a consequence, comparing this virus’s evolution with other endemic viruses, Kathryn Kisler and Trevor Bedford just published, SARS-CoV-2 is accumulating mutations with amino acid substitutions faster than other endemic viruses, including 2.5 fold more rapidly than influenza (A/H3N2 HA1), the prototype of rapid antigen evolution, and >7-fold faster other coronaviruses (Figure below).
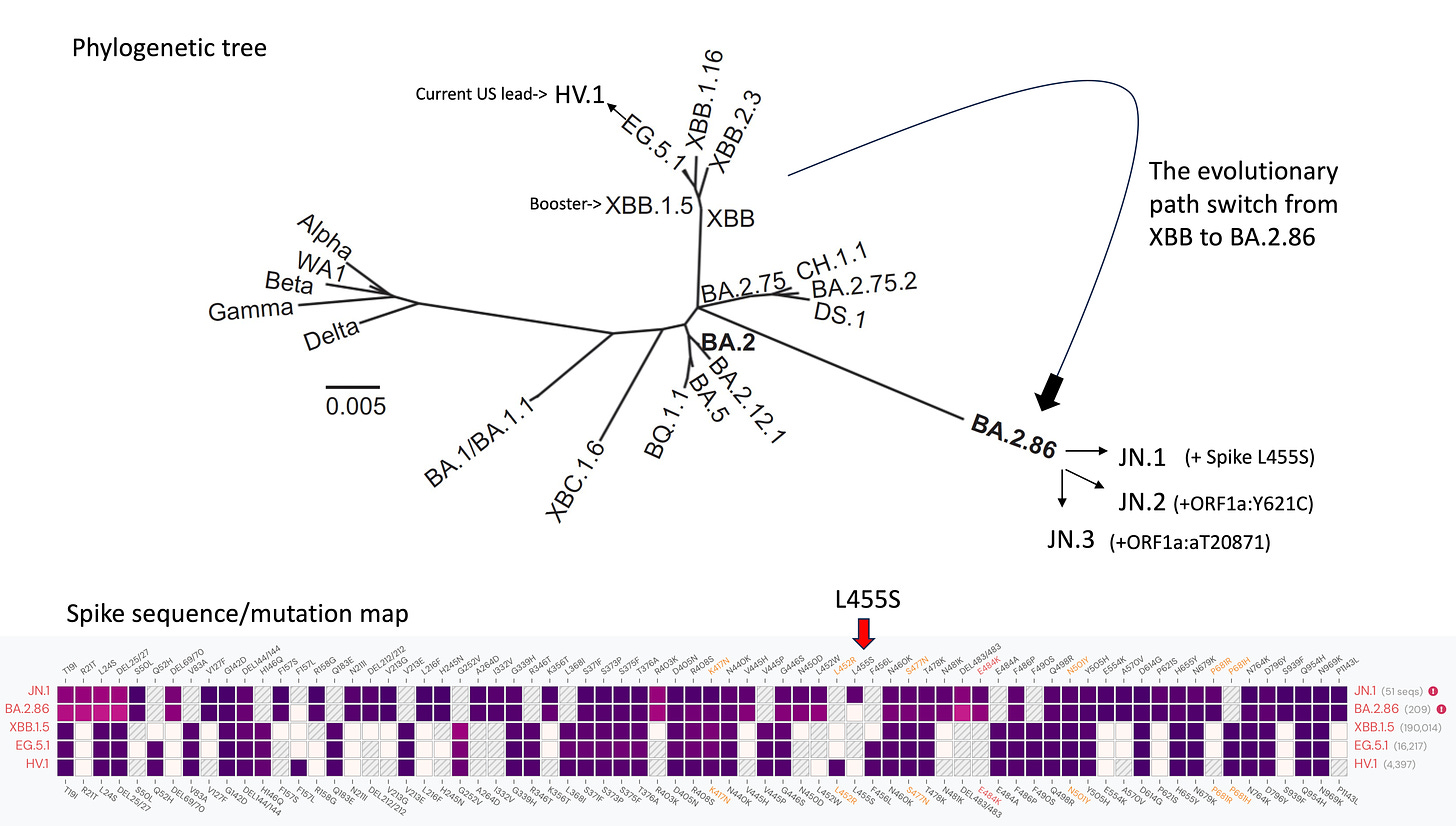
The Figure below maps out what appears now to be taking place. Currently, in the United States, HV.1 is the leading variant (among many others circulating) linked to the new Covid infections. It is a descendant of EG.5.1 and the family of recombinant XBB variants. Recall that the new boosters (Pfizer, Moderna, Novavax) are monovalent, directed specifically to the XBB.1.5 spike. XBB.1.5 predated the rise of EG.5.1 and HV.1, but all are in the XBB tree limb.
But now we are seeing the hyper-mutated BA.2.86 variant (known as “Pirola” in some circles), that came on the scene in August 2023 and I previously reviewed here, which carries >40 new mutations (34 in the spike protein alone) from its BA.2 ancestor, be the new path (a detour, if you will) with addition of the L455S mutation to be known as JN.1.
Why is this potentially important?
The spike mutation map above shows that our booster XBB.1.5 closely resembles EG.5.1 and HV.1, only off by a few mutations. That aligns with a lack of rise in wastewater surveillance or Covid related hospitalizations in recent weeks—that population immunity, along with boosters, is keeping us in a relatively quiescent phase.
But the lab data coming out for properties of BA.2.86 and particularly JN.1 are concerning. The Columbia University Ho lab report in Nature this week reinforced others for the very high affinity that BA.2.86 has towards the ACE2 receptor. which could lead to enhanced transmissibility. Yunlong Cao’s group in Peking was the first to characterize the JN.1 variant, and showed a weak neutralizing antibody response (y-axis, NT50) to breakthrough infections (BTI) of XBB (figure below).
And the impact of the L455S mutation on Class 1 monoclonal antibodies, that differs from BA.2.86 as shown here. This mutation is key to promoting more evasion to our immune response. The FLip variants, which are an ongoing “double whammy mutation” (reviewed here) (involving spike sites 455 and 456) are superseded with respect to immune evasion by JN.1.
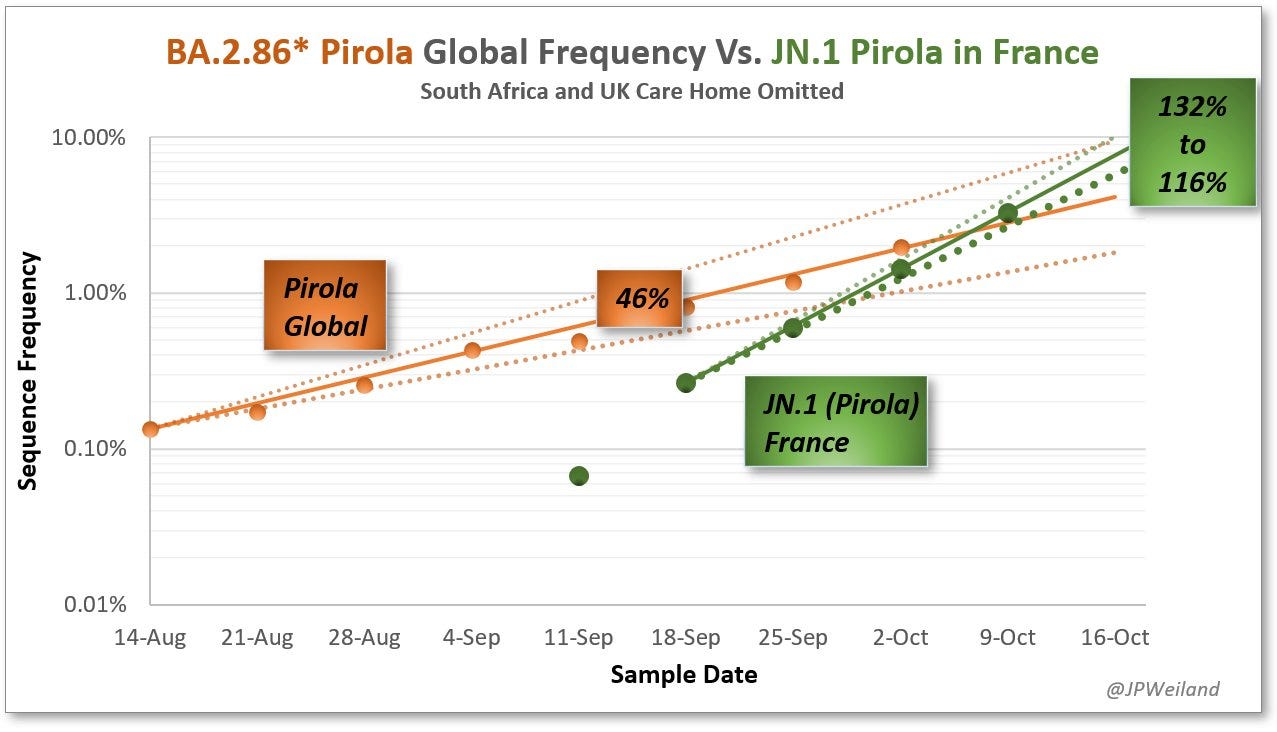
This all seems to be linked with the takeoff of JN.1 in France and other countries. JP Weiland’s model compares BA.2.86 globally with what has been seen for JN.1 in France, suggesting a marked growth advantage of the latter. Nick Rose has also done modeling based on the sequences of JN.1 in France and England with a lesser but still high magnitude of growth advantage.
JN.1 has shown up in many countries now, besides France and the UK, including the US, Iceland, Portugal, Belgium, Israel, Spain, Netherlands, Canada Germany, and Singapore. Other derivatives of BA.2.86 such as JN.2 and JN.3 (see top Figure of post) are also being identified in multiple countries.
We won’t know for a few weeks as to whether JN.1 will be linked with a significant rise in Covid or how well our immune response from prior vaccinations, infection(s) and the XBB.1.5 new booster will keep us protected.
Bottom Line
A detour for the virus’s evolutionary arc appears to be in the works. If this continues, it signifies further distance of the antigen (spike protein) as mapped below by Cao’s team, from where the pandemic started (close to D614G) and where we’ve been recently (EG.5.1 and HV.1). Further away (to JN.1 territory) means it’s harder for our immune system to recognize the antigen— the concept of immune escape.
As I’ve stressed innumerable times previously, we need better vaccines (nasal and pan-coronavirus) to ante up vs the relentless evolution of the virus—its ability, now under population immunity pressure, to keep finding news ways to evade our immune response, enhance transmissibility, and achieve more (re-)infections.
In the meantime, if you haven’t gotten a booster, it would be a very good idea to go ahead. That’s especially important if you are of older age or are immunocompromised. In the U.S. they are approved for all age groups 6 months and older, and there would be some protective benefit across the board, but as with any intervention, a bigger bang for those at highest risk. Even if our current booster does not evoke a strong neutralizing antibody response to JN.1 (or subsequent BA.2.86 descendants), it will rev up our immune system, including cellular immunity, for conferring enhanced protection. As always, non-pharmacologic means of preventing infections help work against all strains of this and other respiratory viruses that are out there now (including flu and RSV).
Even if it turns out that JN.1 is not particularly deleterious, the main message here is that concerning variants keep cropping up and there’s a new path for the virus to find its way—versions that are getting further away from where this all started nearly 4 years ago.
Thanks for reading, subscribing and sharing Ground Truths!






A major hospitality industry trade show just occurred in Las Vegas. A colleague arrived in the US from Europe for the show, didn't feel well, and tested positive for COVID. They, responsibly, remained in their hotel room until testing negative, and missed the entire show. In a conversation with another colleague who knew people who attended came reports of others who came home from the show feeling 'not great' and tested positive. The Show has no contact tracing requirement. As has been the case from the start of COVID, events are not checking to see if hotels' and convention center and other service employees are reporting symptoms of positive tests. It would be great if events do occur they announce "We're a mask-friendly event. Ensure others' safety and your own." Our household is still masking, even in our high-rise, to use the elevator to get the mail even tho' we are fully up-to-date on vaccinations.
I received COVID vaccine recently and three weeks later got COVID. Was fairly sick but seem to be recovering. First time with COVID. Had all other COVID vaccines and boosters and had been pretty careful with masking. Doctor thinks I got one of newer variants that escaped vaccine. This thing is definitely not over.