The Missing CHIP
Ignoring a very informative lab test
It took 8 years for the cardiology professional organizations to revise their guidelines to align with the evidence-based lipid panel, published this week, finally incorporating lipoprotein(a) —Lp(a) and Apolipoprotein B—ApoB. Think of how many people would have been identified at high-risk for atherosclerosis and cardiovascular events who were missed all this time because the guidelines didn’t keep up with the body of knowledge.
Nearly 2 years ago here in Ground Truths (GT) I wrote “The Under-Appreciation of CHIP” calling out the need to take this assay seriously because it conveyed substantial risk information, independent of traditional factors, for cardiovascular disease, cancer, blood clots, and other conditions. If you are interested in this topic (and I hope you will be), I suggest you go back and read that post because I won’t review much of the background here, just a little. In this edition of GT, I’m going to summarize substantial new data on CHIP, review my results, and call for action.
Quick Background
CHIP is the acronym for clonal hematopoiesis of indeterminate potential. It refers to the acquired (somatic) mutation of a blood stem cell that gives it fitness for clonal expansion, with a clone size abnormality cutoff defined as 2% or greater of blood cells, or specifically a variant allele frequency (VAF) ≥ 2% of a white blood cell driver mutation.
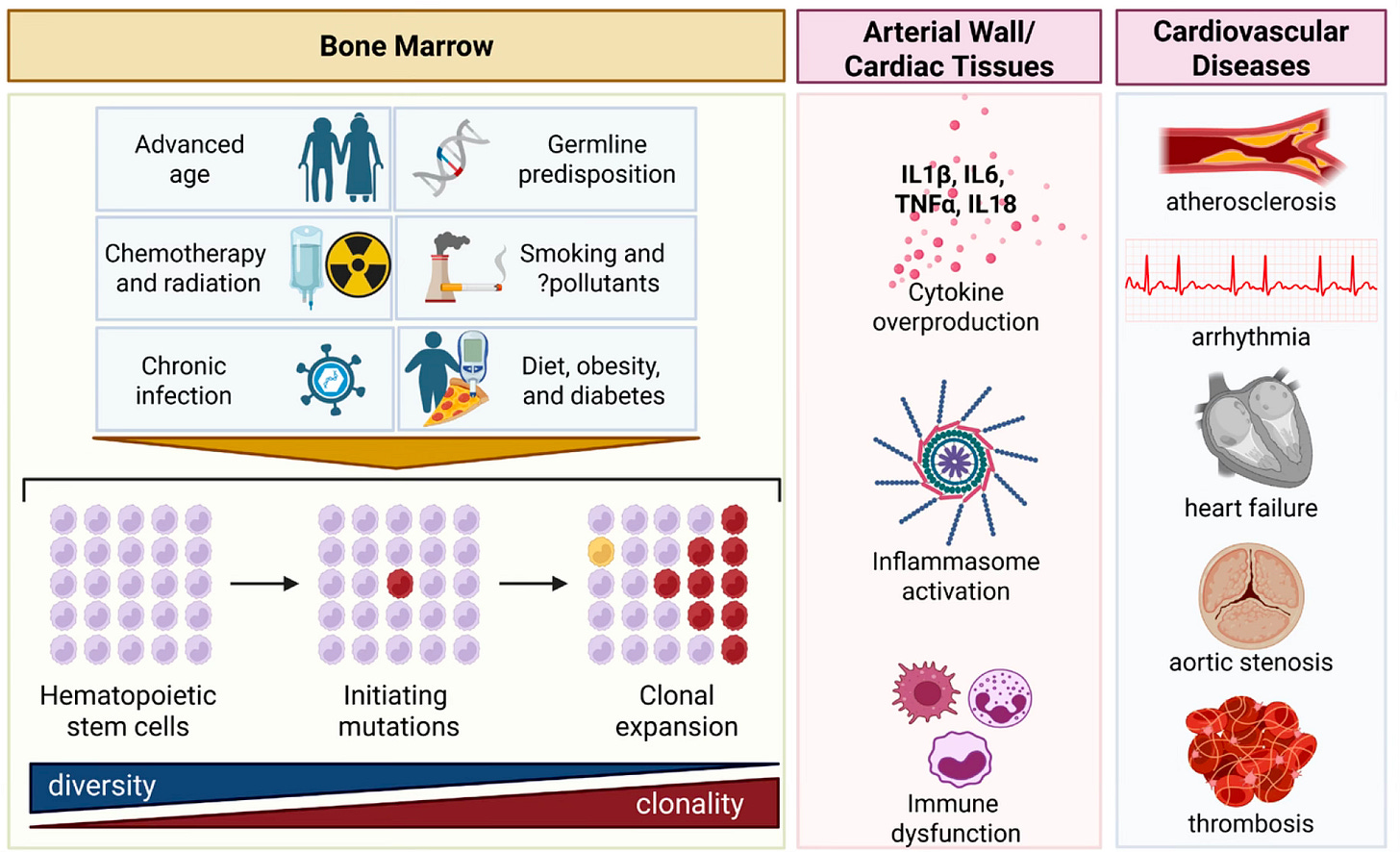
CHIP increases with aging: only 2% of people age 50-69, 10% age 70-79, 20% age 80-89, and >39% age 90+. The most common mutations are DNMT3A, about 48%, TET2, about 22%, ASXL1 11%, and a group of others (such as JAK2, TP53, SF3B1) at 19%. The 3 most common are mutations in epigenetic (methylation side chain, chromatin, regulator) genes.
The presence of CHIP is associated with many disease, especially cardiovascular, cancer, and inflammatory conditions, and the risk increases with higher CHIP frequency.
Last month the American Heart Association published a scientific statement about CHIP: “CH is increasingly recognized as a potential driver of cardiovascular disease.”
Below is the key Figure from that review. Besides advanced age there are other risk factors for developing CHIP. Whit CHIP ≥ 2%, the link with higher levels of inflammation and immune dysfunction, in the context of advanced age, referred to as inflammaging and immunosenescence, is seen. As is the association to multiple cardiovascular diseases and blood cancers. While only 0.5 to 1.0% of people with CHIP > 10% will develop a blood cancer each year, the dysfunction of the immune system and a chronic pro-inflammatory state are detected without disease manifestation. In patients with advanced solid cancers, over 25% have CHIP, much higher than age-matched healthy control individuals.
As reviewed in my previous post on CHIP, the cardiovascular disease hazard ratio for CHIP of 1.8 which exceeds smoking, hypertension, and total cholesterol >200 mg/dl (all at 1.4). A new CHIP risk score has recently been proposed that adds to CHIP status by partitioning CHIP level of risk.
New CHIP Findings
I’ll divide these into disease and treatment related reports, and point out this is not an exhaustive review but a sampling of new studies that strongly reinforce the importance of CHIP.
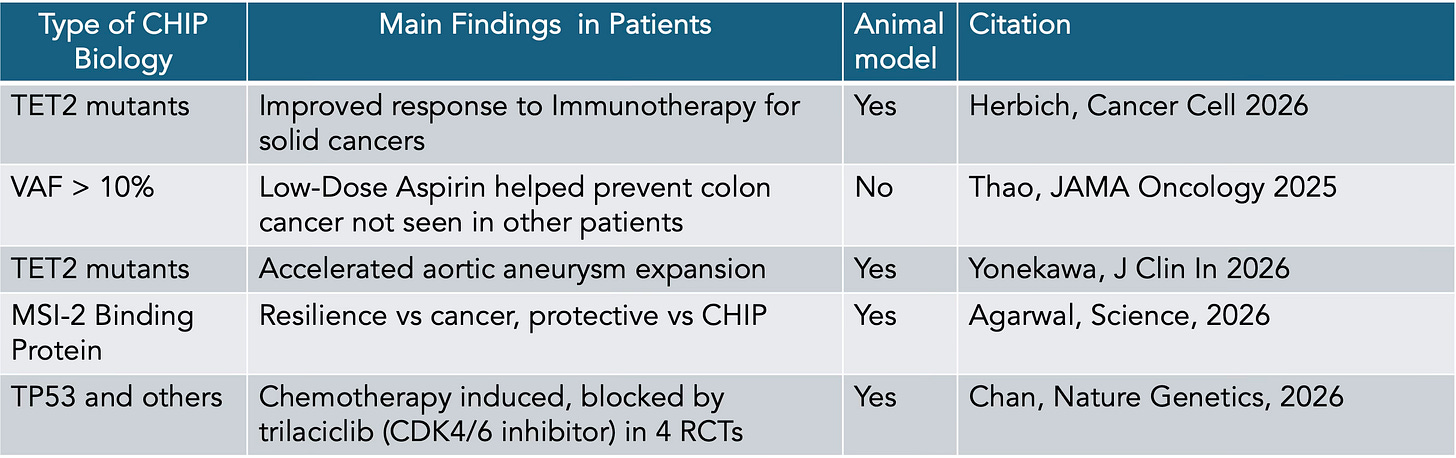
Here’s a Table of some recent disease related findings:
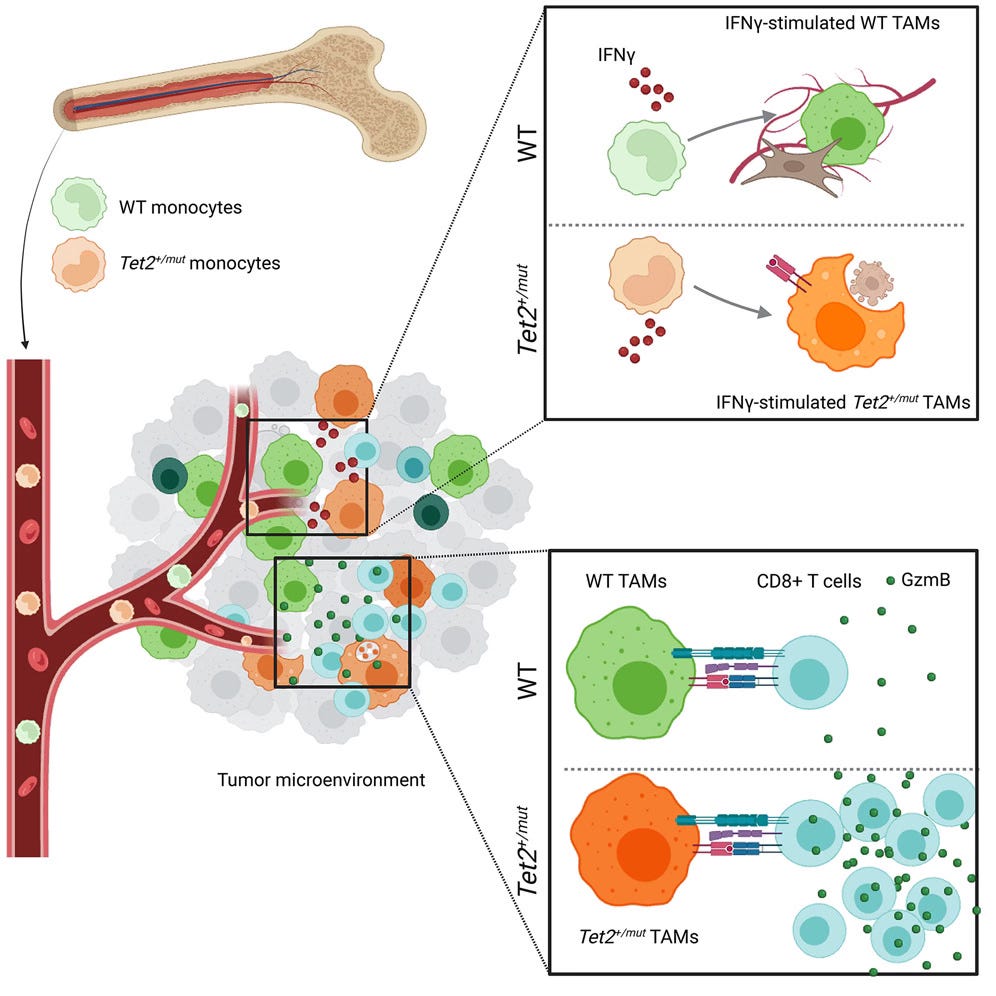
Let me provide a bit of color for these. For improved response to checkpoint therapy for non small cell lung cancer and colorectal cancer, tagged to TET2 mutations, the mechanism was probed below. Note that circulating CHIPs penetrate the tumor microenvironment and here TET2 mutant clones facilitate increased interferon and T-cell response.
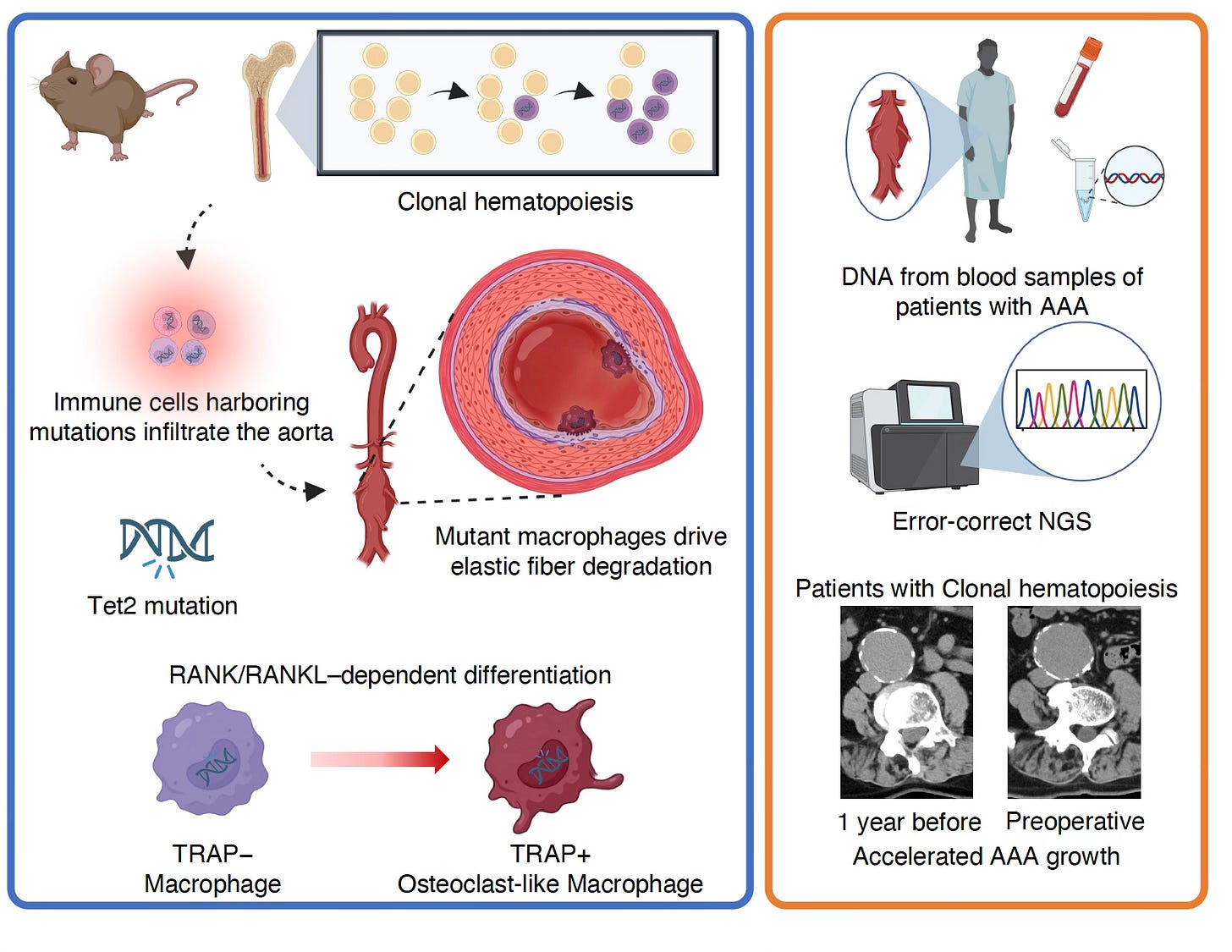
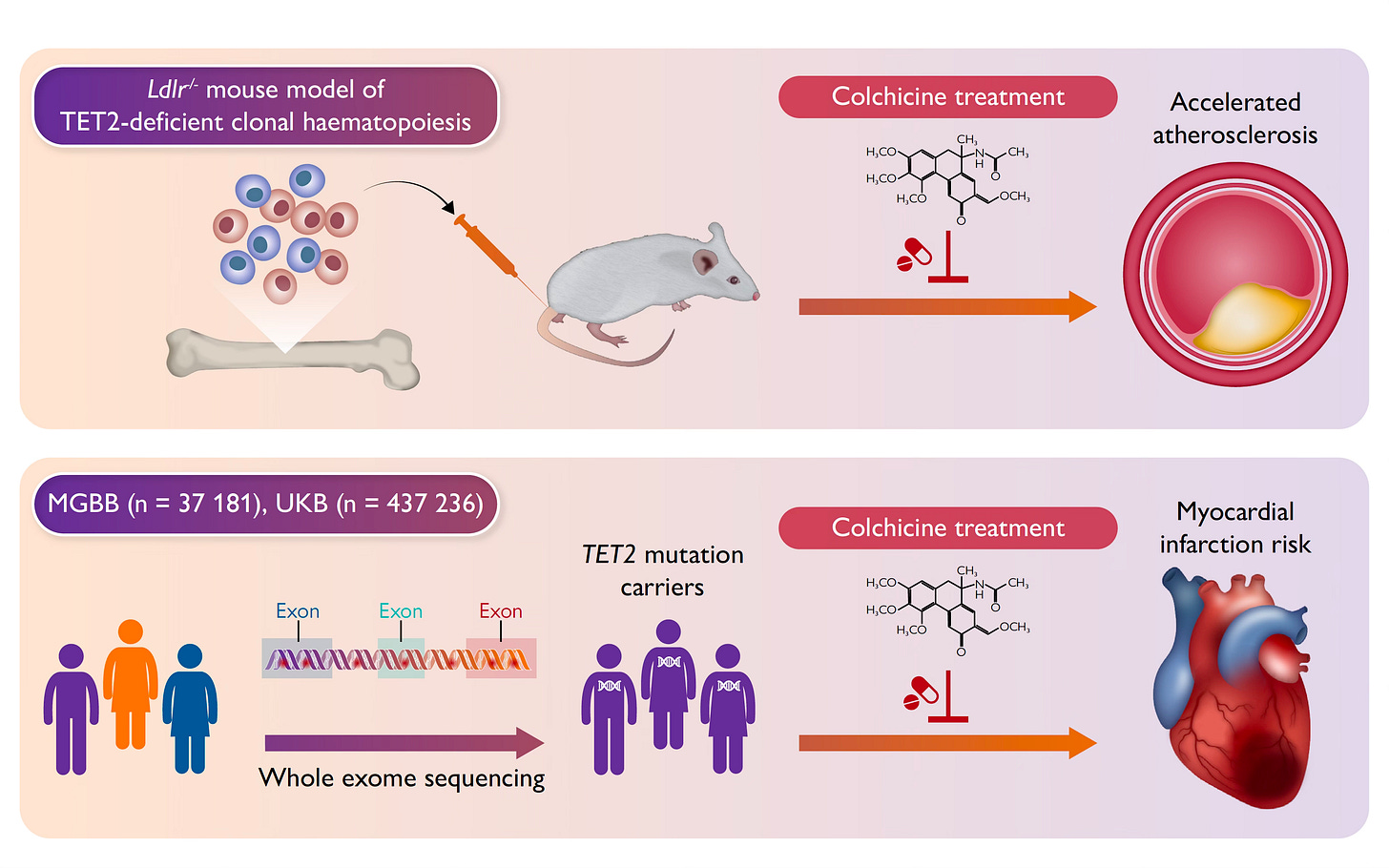
But in a different study of TET2 mutant CHIP, in the presence of an aortic aneurysm, the pro-inflammatory macrophage effect was linked to accelerated aneurysm growth.
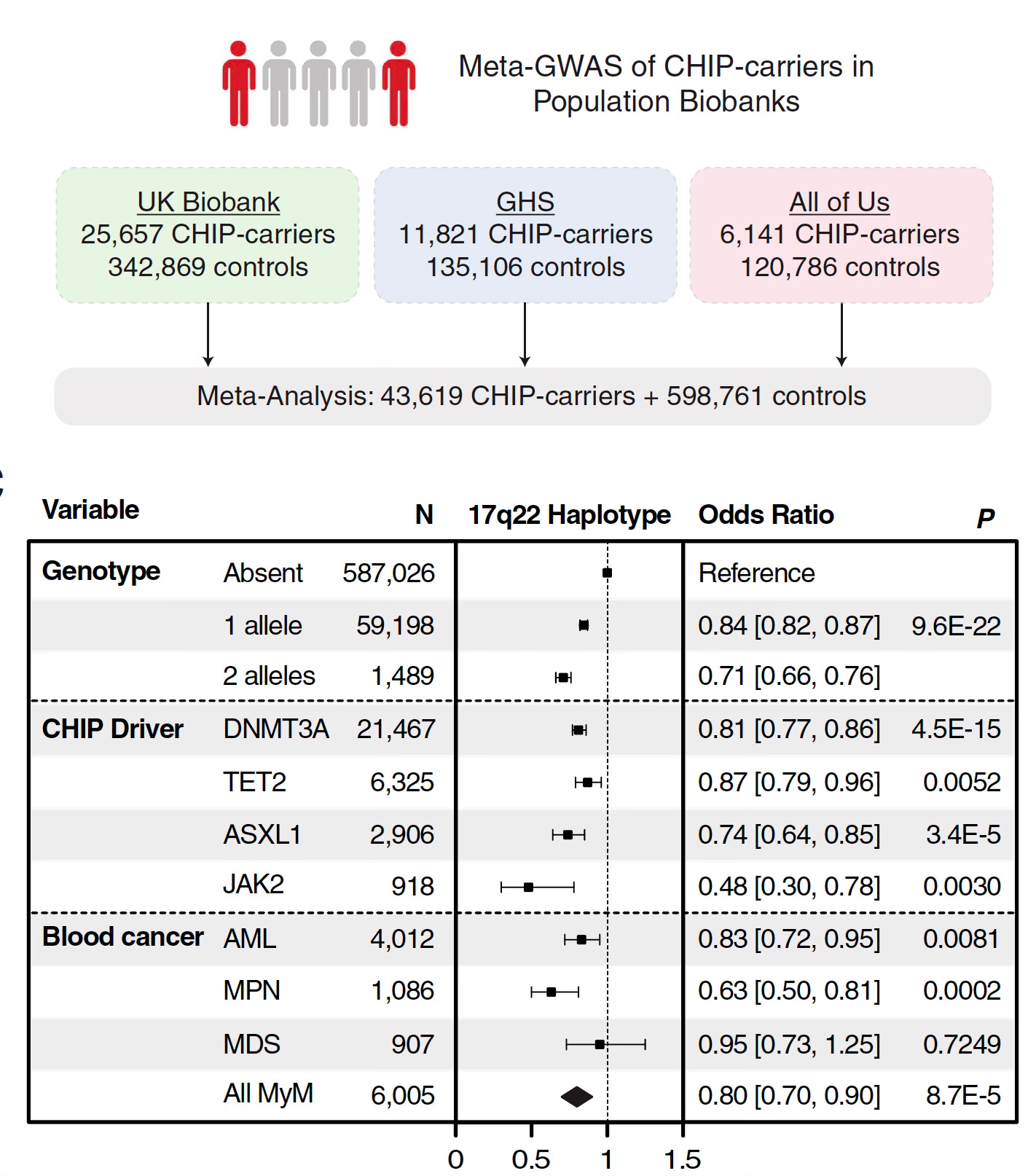
An important discovery for resilience to CHIP, protecting against its formation and related blood cancers, was the discovery of a gene variant on chromosome 17 in 3 large cohorts (Figure) linked to reduction of multiple subtypes of CHIP (driver mutations) and blood cancers. This turned out to be related to an RNA network and specifically down-regulation of the RNA binding protein MSI-2. This seminal finding could lead to a new intervention to suppress CHIP and its adverse health outcome impact.
Now let’s move on to some new treatment findings.
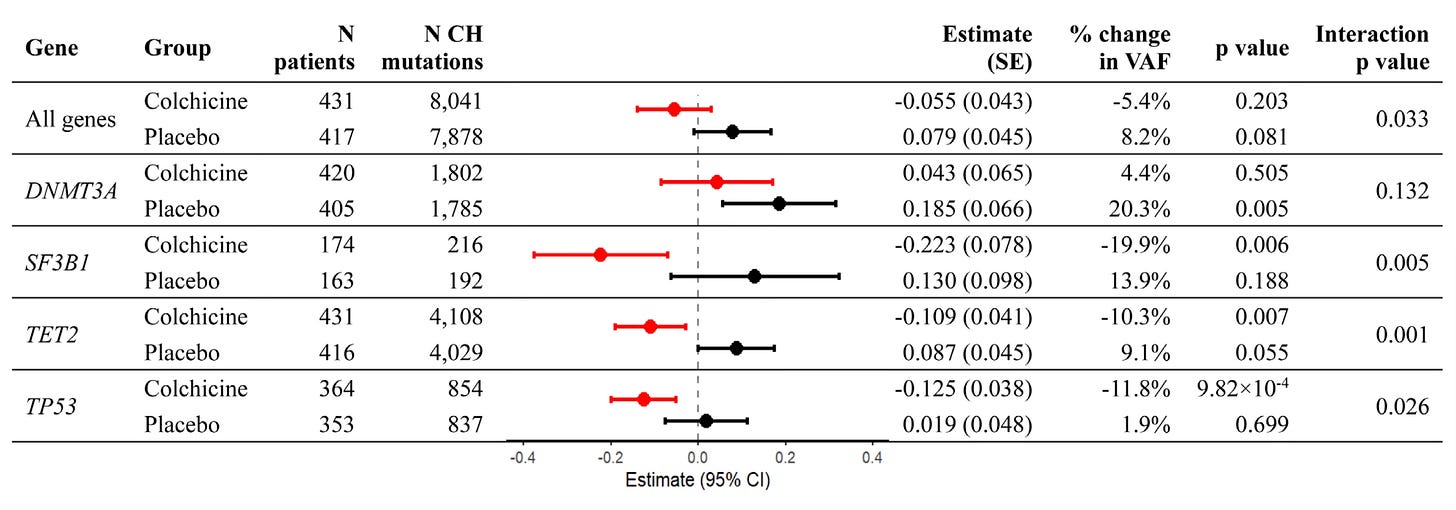
Low dose colchicine was shown to have a consistent effect of reducing CHIP in a randomized placebo-controlled trial with longitudinal sampling (Table).
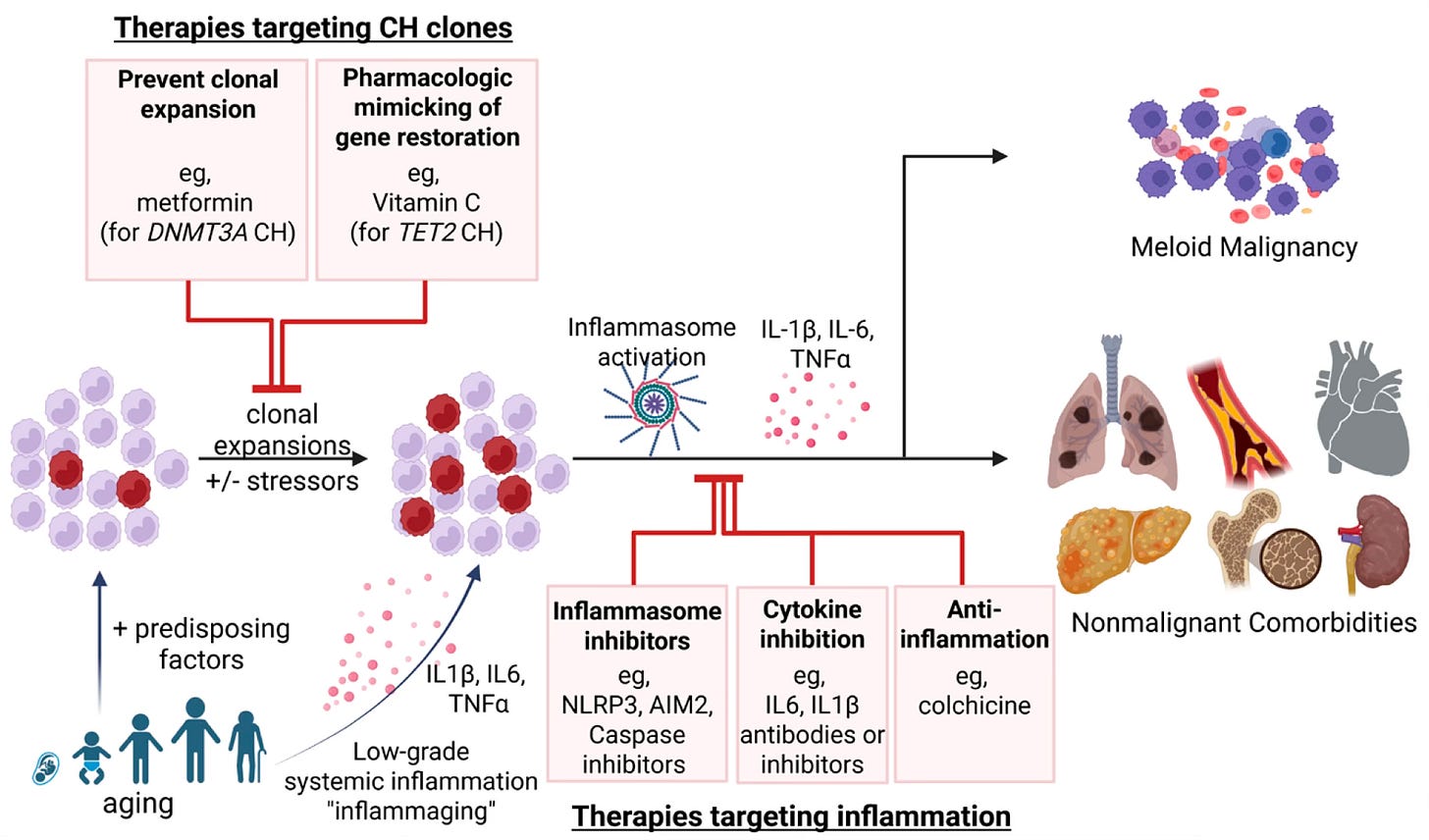
Beyond colchicine (an NLRP3 inflammasome blocker), the IL-1β–neutralizing antibody was shown to reduce events in participants with TET2 in a randomized trial and other potential interventions have been reported such as metformin (vs DNMT3A), Vitamin C (vs.TET2), inhibitors to other interleukins such as IL-6, or inflammasome blockers (Figure). The NLRP3 inflammasome inhibitor selnoflast is being assessed in an ongoing clinical trial for CHIP. Even GLP-1 drugs, known to have portent anti-inflammatory action, have recently been raised as a potential effective anti-CHIP intervention. I should mention that low-dose colchicine, which has the best dataset to date, is remarkably cheap, pennies per day, generic, and well tolerated.
My CHIP Results
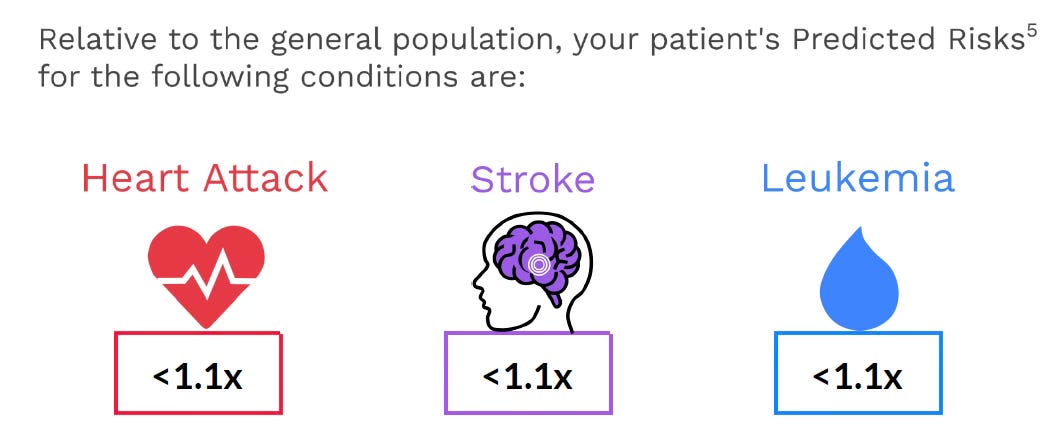
In response to my post on CHIP a couple of years ago, a company (Lucence) offered to provide my CHIP results free of charge. I recently had the blood test and got back the results. My CH was 0.3%, well below the 2% threshold for diagnosis and the only driver mutations detected were in TET2 (4 different variants each at very low VAF). Compared with the general population, not adjusted by age, the result indicated a lower risk for the 3 major outcomes as shown below. In my age group, the chance of having CHIP (defined as >2%) is more than 10%, so I feel fortunate. Had I been found to have TET-2 CHIP I might have considered starting low-dose colchicine.
The Implications
We’re learning much more about CHIP, how it links to a risk of many diseases spanning cardiovascular, cancer, thromboembolic (clotting) and liver (previously reviewed in GT). Different driver mutations carry different risks, and even the same CHIP mutations, such as TET2, can do both harm (aneurysm growth) and good (improve response to soldi tumor immunotherapy). The fact that these are multiple clinical studies fully backed up by animal models supports evidence for causality. Some mutations, like DNMT3A, have been linked to improved survival in colon cancer, so the presence of CHIP is not always a bad sign. While JAK2 is an unusual subtype of CHIP (range 0.5 to 4% of CHIP driver mutations), it is specifically associated with clotting complications, such as pulmonary embolism.
The CHIP cause and effect relationship is not yet considered fully established but the new data from multiple parallel human and animal studies strongly support that CHIP is not just a link to adverse outcomes, that it is highly likely to be a driver.
So why don’t we have routine CHIP assays for people of older age? One reason would be we don’t have therapies that are effective, but that seems to be negated with low- dose colchicine and many other candidate drugs with promising data for effectiveness (either blocking CHIP formation or its downstream risk of diseases). Another reason is that CHIP is hard to assay accurately. While it can be detected by whole genome or whole exome sequencing, the gold standard is to use a narrow, targeted panel of ~100 genes and do very deep sequencing, exceeding 500X. That is what was used by Lucence to do my assay, but it has a charge of $950 which is extremely high and I consider untenable. With the marked reduction in sequencing costs in the last couple of years, getting a precise and accurate CHIP assay at low cost should be a priority, in conjunction with dedicated clinical trials of drugs that prove outcome improvements.
Another potential downside of CHIP lab tests is inducing anxiety, such as seen in cancer survivors, and others with high-risk results for major diseases. But the trade-off of close surveillance of such individuals and mitigation of risk factors deserves consideration. Getting ahead of a disease enables prevention, which is typically far better than resorting to a treatment.
With all this background, it is notable that there are currently 2 academic centers that have established dedicated CHIP Clinics, both the University of Chicago and Cleveland Clinic. After my initial post, I learned from Dr. Jennifer Kwan there is a hybrid cardio-oncology CHIP clinic at Yale Medicine. Why aren’t there more? With all the evidence that keeps accumulating, why aren’t we making CHIP assessment widely available at low cost for select individuals (such as 70+ years of age)? Our blood makers for inflammation, such as high-sensitivity C-reactive protein, are notoriously non-specific and can be misleading. I would like to change the CHIP acronym to Clonal Hemaotopoiesis of Important Potential, getting rid of the Indeterminate qualifier. It is certainly informative. I think more so than most of the hundreds of tests that companies like Function Health provide to their several hundred thousand customers. Like Lp(a), will it take another 8 years before we have new guidelines from cardiovascular, hematology and oncology professional societies that back up the utility of diagnosing and treating CHIP?
NB: I wrote this post with no AI assistance. I have no relationship with any company that does the CHIP assay or potential treatments that would treat CHIP.
**********************************************
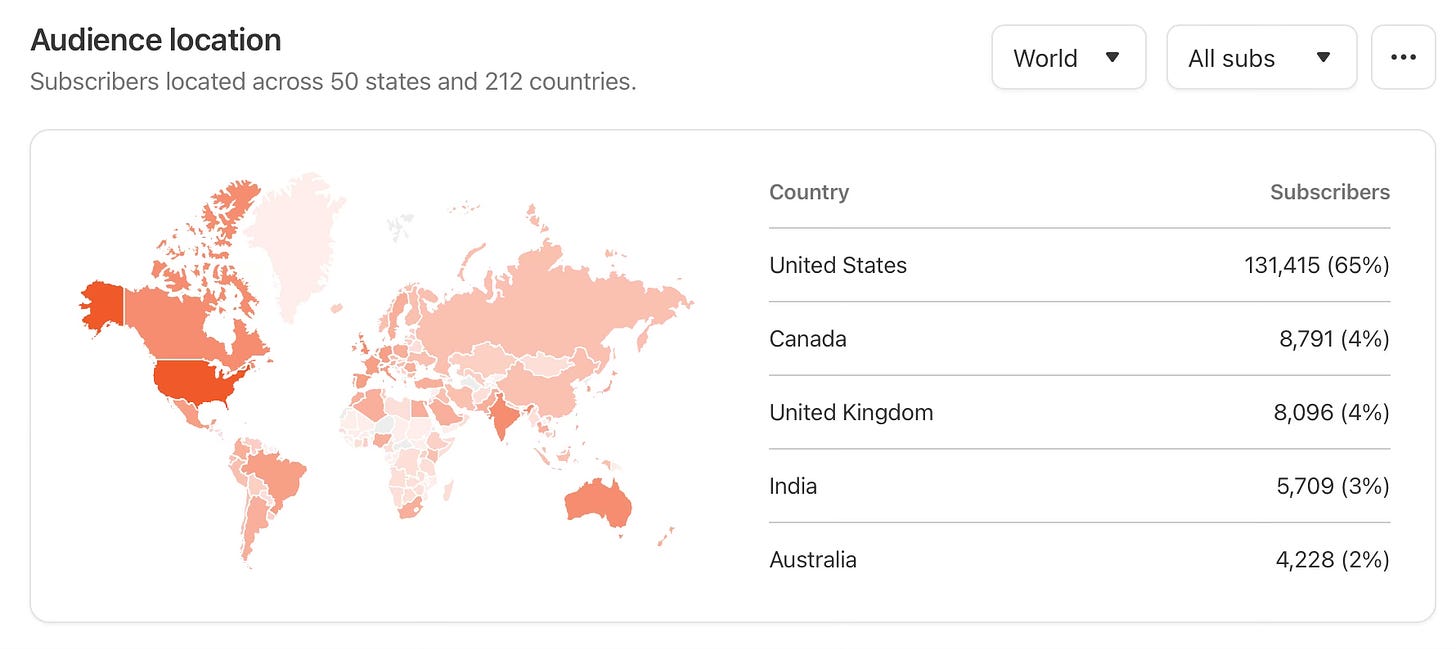
Thanks to Ground Truths subscribers (now > 200,000) from every US state and 212 countries. Your subscription to these free essays and podcasts makes my work in putting them together worthwhile. Please join!
If you found this interesting PLEASE share it!
Paid subscriptions are voluntary and all proceeds from them go to support Scripps Research. They do allow for posting comments and questions, which I do my best to respond to. Please don’t hesitate to post comments and give me feedback. Let me know topics that you would like to see covered.
Many thanks to those who have contributed—they have greatly helped fund our summer internship programs for the past two years. It enabled us to accept and support 47 summer interns in 2025! We aim to accept even more of the several thousand who will apply for summer 2026.

Thanks for highlighting this marker. I have been monitoring mine for a while but did not know until now that there are interventions.
WRT "low-dose colchicine" what sort of dose are we talking about? Are there non-prescription alternatives? Should this be recommended for everybody until the CHIP test becomes cheaper?